
GCEM provides design reflected by GMP regulations including FDA, EMEA, WHO, PIC, etc. through business experiences with GMP Consulting and Engineering companies in the world. It also provides design on the best condition by selecting the optimum system based on customers’ demands including BL4, BL3, research facilities, health examination centers, vivarium, food, cosmetics, etc.
-
Concept

In the conceptual design phase, we confirm the layout via adjustment and arrangement of required rooms in consideration of GMP flows and production capacity.
- Project Management
- Mechanical Concept
- Clean Utility Concept
- Architecture Concept
- Electrical Concept
- Process Concept
-
Basic

The Basic Engineering phase includes time schedule, expenses and details of the results of Conceptual Design.
- Review of Concept Design
- Project Master Plan (PMP)
- Review of production facilities and equipment
- Description and planning of building
- Confirm Lay-out
- System P & IDs (Process, Utility)
- Load calculation
- Expenses and schedule (± 15%)
-
Detail

The Detail Engineering phase includes detailed working drawing, detailed expenses and construction schedule.
- Review of basic design
- Detailed design plan and detailed statement
- Review report to purchase equipment
- Specification/Various bills
- VE report
- Final budget
- Construction schedule
-
BIM
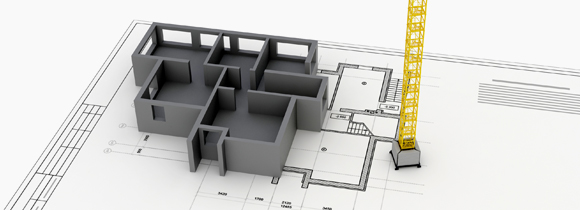
Checking error and interference of design by 3D modelling 2D design Detail Design through BIM prevents problem occurrence due to error and supports project decision making by correctly calculating expenses and schedule.
- Process schedule through BIM model
- Quotation and quantity estimation through BIM model
- Simulation video (air current, temperature and moisture, differential pressure)
